Facile synthesis of porous iron oxide/graphene hybrid nanocomposites and potential application in electrochemical energy storage†
Abstract
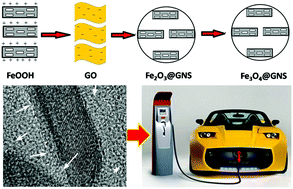
A facile and efficient method is used to synthesize porous iron oxide coated with graphene as electrode materials for lithium-ion batteries and supercapacitors. Graphene encapsulation of porous Fe2O3 and Fe3O4 nanorods is directly carried out from FeOOH@GO colloids by taking advantage of an electrostatic self-assembly method, owing to the positively-charged surface of FeOOH and the negatively-charged surface of GO. The combination of graphene and porous iron oxide brings about multifunctional features of the electrode materials as follows: (1) enhanced electrical conductivity makes the electrodes the current collectors; (2) reinforced softness of the electrodes accommodates the large volume changes during charge–discharge cycles; (3) improved high specific surface area of the electrodes increases the accessibility of the active electrode materials to electrolyte; (4) the pores formed by graphene and iron oxide particles facilitate ion transportation; (5) iron oxide particles separate graphene and prevent their restacking or agglomeration, and vice versa, thus improving the immersion and splitting of electrolyte into and out of the electroactive material. Consequently, the porous iron oxide/graphene hybrid nanocomposites deliver a good performance in the electrochemical energy storage for lithium-ion batteries and supercapacitors.


 Please wait while we load your content...
Please wait while we load your content...