Decomposition behavior of platinum clusters supported on ceria and γ-alumina in the presence of carbon monoxide
Abstract
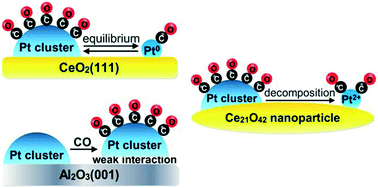
Since the catalytic behavior of supported platinum species strongly depends on their nuclearity under the reaction conditions, in this study we addressed the stability of platinum species in the presence of CO. We applied density functional modeling to clarify the effect of the CO coverage on the structure and stability of small platinum clusters deposited on CeO2(111) and γ-Al2O3(001) surfaces and on a ceria nanoparticle. The stability was evaluated with respect to decomposition of the clusters to Pt, Pt0(CO) and/or Pt2+(CO)2 species. The results suggest that decomposition of pristine platinum clusters to Pt0 atoms is endothermic on all considered supports. On the other hand, the formation of Pt0(CO) monocarbonyls is essentially energy-neutral when the platinum cluster deposited on the ceria nanoparticle or ceria (111) surface is fully covered by CO molecules. Formation of Pt2+(CO)2 species can occur only when the support is a small ceria nanoparticle which ensures low-coordinated O centers and easily reducible Ce4+ ions. On the γ-Al2O3(001) surface, the platinum cluster remains stable with respect to decomposition even when it is fully covered by CO but the interaction of the cluster with the alumina surface is strongly weakened. Thus, based on the results, one may suppose that under CO pressure platinum clusters behave differently depending on the support – decomposition to neutral monocarbonyls on the ceria surface and to cationic complexes on the ceria nanoparticle, while on alumina the carbonylated cluster remains intact but is almost detached from the surface.


 Please wait while we load your content...
Please wait while we load your content...