The nature of selenium hydrogen bonding: gas phase spectroscopy and quantum chemistry calculations†
Abstract
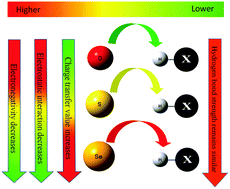
Subsequent to the recent re-definition of hydrogen bonding by the IUPAC committee, there has been a growing search for finding the presence of this ever interesting non-covalent interaction between a hydrogen atom in an X–H group and any other atom in the periodic table. In recent gas phase experiments, it has been observed that hydrogen bonding interactions involving S and Se are of similar strength to those with an O atom. However, there is no clear explanation for the unusual strength of this interaction in the case of hydrogen bond acceptors which are not conventional electronegative atoms. In this work, we have explored the nature of Se hydrogen bonding by studying indole⋯dimethyl selenide (indmse) and phenol⋯dimethyl selenide (phdmse) complexes using gas phase IR spectroscopy and quantum chemistry calculations. We have found through various energy decomposition analysis (EDA) methods and natural bond orbital (NBO) calculations that, along with electrostatics and polarization, charge transfer interactions are important to understand Se/S hydrogen bonding and there is a delicate balance between the various interactions that plays the crucial role rather than a single component of the interaction energy. An in-depth understanding of this type of non-covalent interaction has immense significance in biology as amino acids containing S and Se are widely present in proteins and hence hydrogen bonding interactions involving S and Se atoms contribute to the folding of proteins.
- This article is part of the themed collections: PCCP Emerging Investigator Lectureship Award Winners and 1st International Conference on Noncovalent Interactions


 Please wait while we load your content...
Please wait while we load your content...