Antiscaling efficacy of CaCO3 and CaSO4 on polyethylene glycol (PEG)-modified reverse osmosis membranes in the presence of humic acid: interplay of membrane surface properties and water chemistry†
Abstract
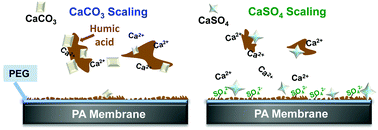
Mineral scaling persists in many water treatment processes. More specifically, it can significantly reduce the efficacy of aromatic polyamide (PA) membranes during reverse osmosis (RO) water treatment. Previous studies have integrated hydrophilic materials, such as polyethylene glycol (PEG), onto RO membranes to combat scaling from generally hydrophobic feed water constituents; however, there are still outstanding knowledge gaps regarding the interplay of the modified membrane surface chemistry and the water chemistry in complex RO feed waters. In this work, we have investigated the mechanisms of hydrophilic PEG-grafted PA membranes in reducing mineral scaling from calcium carbonate (CaCO3) and calcium sulfate (CaSO4) in the presence of humic acid (HA). Based on surface and solution analyses, we found that colloidal formation was significantly reduced on PA–PEG surfaces in systems without HA. When HA was introduced, CaCO3 scaling was reduced on both virgin and PA–PEG membrane surfaces; while, interestingly, synergistic PEG–HA–CaSO4 interactions increased CaSO4 colloidal formation on PA–PEG membranes. Promoted CaSO4 formation results from a high negative surface charge near the PEG-modified membrane surface when HA and SO42− are present, attracting more Ca2+ to form CaSO4. The results of this work provide new information about colloidal formation at water–membrane interfaces for designing better PEG and PEG-based scale-resistant desalination membranes.


 Please wait while we load your content...
Please wait while we load your content...