Solution-based sequential modification of LiCoO2 particle surfaces with iron(ii) oxalate nanolayers†
Abstract
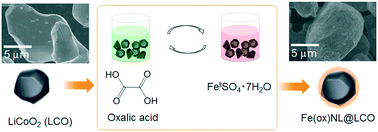
Hybridization of metal–organic frameworks (MOFs) with other inorganic materials offers the opportunity to enhance their properties or open new windows for potential applications. Formation of core/shell structures, in which the core is the inorganic particle and the shell is the surface-deposited MOF, is one of the rational methodologies to create such hybrid materials. Here, we present the facile creation of metal oxide-core/MOF-shell structures by applying a solution-based step-by-step modification of the core surface using a framework compound. The modification procedure includes alternate immersion of LiCoO2 (LCO) – a catalyst for the oxygen evolution reaction – into oxalic acid and ferrous solutions, resulting in the formation of iron(II) oxalate (Fe(ox), ox = oxalate anion) – one of the simplest MOFs with short unit frameworks – nanolayers on the LCO surface (Fe(ox)NL@LCO). The amount of deposited Fe(ox) does not increase monotonically with the number of the modification cycles, but tends to saturate rapidly, implying that the Fe(ox) framework grows laterally on the LCO surface. An increase of the electrical resistivity of the LCO particles that influences adversely catalytic activity is suppressed when a high coverage of the LCO surface by the Fe(ox) framework is achieved at subsequent modification cycles.
- This article is part of the themed collection: Metal-Organic Framework Catalysis


 Please wait while we load your content...
Please wait while we load your content...