Mononuclear gold(iii) complexes with l-histidine-containing dipeptides: tuning the structural and biological properties by variation of the N-terminal amino acid and counter anion†
Abstract
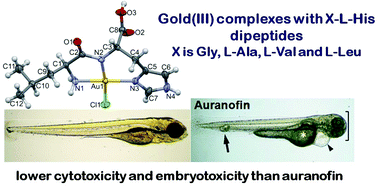
Gold(III) complexes with different L-histidine-containing dipeptides, [Au(Gly-L-His-NA,NP,N3)Cl]Cl·3H2O (1a), [Au(Gly-L-His-NA,NP,N3)Cl]NO3·1.25H2O (1b), [Au(L-Ala-L-His-NA,NP,N3)Cl][AuCl4]·H2O (2a), [Au(L-Ala-L-His-NA,NP,N3)Cl]NO3·2.5H2O (2b), [Au(L-Val-L-His-NA,NP,N3)Cl]Cl·2H2O (3), [Au(L-Leu-L-His-NA,NP,N3)Cl]Cl (4a) and [Au(L-Leu-L-His-NA,NP,N3)Cl][AuCl4]·H2O (4b), have been synthesized and structurally characterized by spectroscopic (1H NMR, IR and UV-vis) and single-crystal X-ray diffraction techniques. The antimicrobial efficiency of these gold(III) complexes, along with K[AuCl4] and the corresponding dipeptides, was evaluated against the broad panel of Gram-positive and Gram-negative bacteria and fungi, displaying their moderate inhibiting activity. Moreover, the cytotoxic properties of the investigated complexes were assessed against the normal human lung fibroblast cell line (MRC5) and two human cancer, cervix (HeLa) and lung (A549) cell lines. None of the complexes exerted significant cytotoxic activity; nevertheless complexes that did show selectivity in terms of cancer vs. normal cell lines (2a/b and 4a/b) have been evaluated using zebrafish (Danio rerio) embryos for toxicity and antiangiogenic potential. Although the gold(III) complexes achieved an antiangiogenic effect comparable to the known angiogenic inhibitors auranofin and sunitinib malate at 30-fold higher concentrations, they had no cardiovascular side effects, which commonly accompany auranofin and sunitinib malate treatment. Finally, binding of the gold(III) complexes to the active sites of both human and bacterial (Escherichia coli) thioredoxin reductases (TrxRs) was demonstrated by conducting a molecular docking study, suggesting that the mechanism of biological action of these complexes can be associated with their interaction with the TrxR active site.


 Please wait while we load your content...
Please wait while we load your content...