Redox behavior of the SOFC electrode candidate NdBaMn2O5+δ investigated by high-temperature in situ neutron diffraction: first characterisation in real time of an LnBaMn2O5.5 intermediate phase†
Abstract
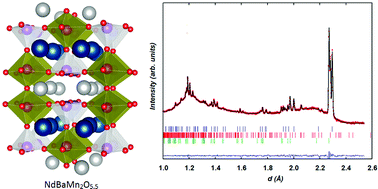
The structural behavior of the tetragonal NdBaMn2O5 phase, a member of the family of A-site ordered layered manganites that have been recently suggested as possible mixed ionic and electronic conductors, has been investigated by means of in situ neutron powder diffraction. Considering applications in energy production and storage devices and use of NdBaMn2O5+δ as an electrode in symmetrical cells, the study was carried out in relevant atmosphere conditions, i.e. dilute hydrogen (wet and dry) and dry air in the temperature range 25–800 °C. Neutron data under flowing hydrogen allowed monitoring of the structural phase transition from the charge-ordered to the charge-disordered state as a function of temperature. Slow reduction of the fully oxidised phase, NdBaMn2O6, previously formed from quick oxidation of the pristine material, enabled real-time observation of the intermediate NdBaMn2O5.5 phase and its crystal characterization up to 700 °C in the course of its conversion to NdBaMn2O5. Oxygen vacancy ordering within the Nd layers of NdBaMn2O5.5 correlated with antiferrodistortive orbital ordering of the Jahn–Teller Mn3+ ion in the square pyramids and octahedra results in large thermal expansion and relatively slow anisotropic oxygen diffusion occurring in the NdO layer. The four heating/cooling cycles evidenced no oxygen miscibility between the three distinct phases detected in the NdBaMn2O5+δ system with δ ∼ 0, 0.5 and 1 and clearly demonstrated that reversible oxygen intercalation/deintercalation underpins the phase stability of the LnBaMn2O5+δ materials to redox cycling and to wet atmosphere in high temperature electrochemical devices.


 Please wait while we load your content...
Please wait while we load your content...