Facile synthesis of highly active and durable PdM/C (M = Fe, Mn) nanocatalysts for the oxygen reduction reaction in an alkaline medium†
Abstract
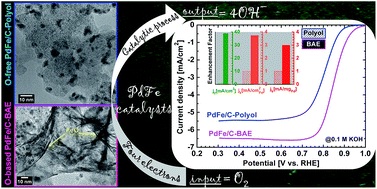
The efficient design of highly active and durable materials towards the ultimate goal of improving kinetics of the oxygen reduction reaction (ORR), which allow enhanced performance in solid alkaline membrane fuel cells (SAMFCs), remains elusive. Seminal studies have shown that by alloying a noble metal such as palladium to a transition metal, it is possible to tune the electronic and/or bifunctional properties enabling substantial ORR performance to be achieved, thereby designing a costly catalyst. Herein, we address and discuss new findings from deeper ORR investigations at palladium-based nanostructures in an alkaline medium. We exploited and manipulated the straightforward and fast synthesis method, the so-called “Bromide Anion Exchange”, to prepare surfactant-free PdM/C (M = Fe, Mn) nanocatalysts exhibiting unprecedented activity and stability towards ORR. PdFe/C from bromide anion exchange (BAE) enables 40- and 4-fold enhancement in terms of exchange current density and kinetic current density and ca. 100 mV gains compared to the polyol microwave-assisted method. After 20 000 cycles of accelerated potential cycling test (APCT), our findings indicate that the present PdM/C bimetallics outperform, to the best of our knowledge, most of the data reported for ORR in alkaline media for Pd-based transition metals. The improved catalytic performances are assigned to the absence of any organic contaminants or protective ligands on their surface and their relatively heterogeneous character comprising nanoalloys and nanowire oxides.

 Please wait while we load your content...
Please wait while we load your content...