Tri-layered graphite foil for electrochemical capacitors†
Abstract
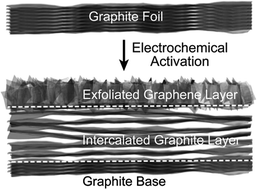
Free-standing carbon structures are promising electrode materials for electrochemical capacitors. However, these electrodes usually have small mass that limits the amount of energy that can be stored. Increasing the electrode mass typically leads to reduction of gravimetric capacitance and rate capability due to the sluggish mass transfer kinetics and increased internal resistance. It has been a challenge to improve both specific capacitance and rate capability of an electrode with high mass. Here we demonstrate a new method to convert graphite foil (8.5 mg cm−2) with a compact layered structure into a unique tri-layered structure that consists of a top layer of partially exfoliated graphene sheets, a middle layer of intercalated graphite sheets and a bottom layer of graphite. This unique structure shows enhanced ion accessible surface area and pseudocapacitance. The seamless connection between the three layers ensures efficient electron transport across the electrode. The tri-layered graphite foil electrode delivers an excellent capacitance of 820 mF cm−2 at 5 mA cm−2 (corresponding to 96.5 F g−1), which is 400 times higher than the untreated foil. Moreover, it retains 75% capacitance when the current density is increased from 5 to 100 mA cm−2. These values are among the best values reported for carbon-based electrodes with comparable mass.

 Please wait while we load your content...
Please wait while we load your content...