Thermodynamic and conformational changes of protein toward interaction with nanoparticles: a spectroscopic overview
Abstract
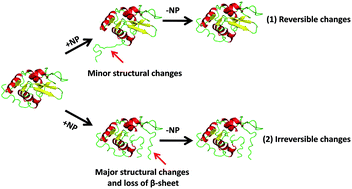
Nanoparticles (NPs) in different forms have been widely used in medicine and pharmaceutics for diagnosis and drug delivery. So, they are in intimate contact with bio-macromolecules like proteins which are vital and functional elements of cells and organisms. Thus, in vitro studies of the possible interaction between proteins and NPs are necessary from both application and toxicity points of view. In this article, binding characteristics and protein conformational change through interaction with various NPs have been reviewed. In recent years, a wide variety of biophysical techniques have been exploited to elucidate mechanistic aspects of NP–protein interactions. This study has focused on the information acquired from prevalent techniques including fluorescence quenching, synchronous fluorescence spectroscopy, circular dichroism (CD), and surface-enhanced Raman scattering (SERS).

 Please wait while we load your content...
Please wait while we load your content...