Synthesis, molecular and electronic structure of a stacked half-sandwich dititanium complex incorporating a cyclic π-faced bridging ligand†
Abstract
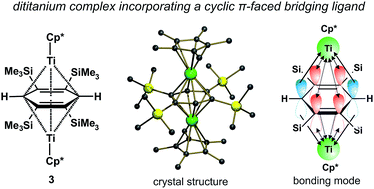
A thermally robust triple-decker complex [bis(η5-pentamethylcyclopentadienyltitanium)-μ-(η4:η4-1,2,4,5-tetrakis(trimethylsilyl)cyclohexa-1-4-diene-3,6-diyl)] (3) was obtained in 4% yield by thermolysing Cp*TiMe3 in the presence bis(trimethylsilyl)acetylene (BTMSA). The solid-state structure of centrosymmetric 3 features rather long all C–C bonds in the nearly planar bridging ligand (1.4720(14)–1.4896(15) Å) and a short distance of its least-square plane to the titanium atoms (1.7381(5) Å). Computational results revealed the bonding of the central ligand to be accomplished through back-bonding of its two C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C bonds and through the simultaneous generation of two σ-Ti–C(H) bonds. Based on CASSCF and CASPT2 results, the molecule acquires several electronic configurations simultaneously, which hinders its representation by one single Lewis structure. Apart from being coordinated to the central ligand, the metal atoms are involved in a direct Ti–Ti bonding by the formation of one σ- and two δ-bonds between them. The bond order of this Ti–Ti overlap shows only a slight decrease upon electronic excitation. The presence of ionic contribution to the bonding of the central ligand is manifested by the charge −1.4e summed on the carbon atoms of the bridging ring. Based on computational results, the spin multiplicity of the ground state is singlet, while the first low lying excitation state is triplet. This is in agreement with the absence of EPR signal in either toluene solution and glass, and with slight downfield shifts of broadened 1H NMR signals of SiMe3 and Cp* methyl groups observed with increasing temperature.
C bonds and through the simultaneous generation of two σ-Ti–C(H) bonds. Based on CASSCF and CASPT2 results, the molecule acquires several electronic configurations simultaneously, which hinders its representation by one single Lewis structure. Apart from being coordinated to the central ligand, the metal atoms are involved in a direct Ti–Ti bonding by the formation of one σ- and two δ-bonds between them. The bond order of this Ti–Ti overlap shows only a slight decrease upon electronic excitation. The presence of ionic contribution to the bonding of the central ligand is manifested by the charge −1.4e summed on the carbon atoms of the bridging ring. Based on computational results, the spin multiplicity of the ground state is singlet, while the first low lying excitation state is triplet. This is in agreement with the absence of EPR signal in either toluene solution and glass, and with slight downfield shifts of broadened 1H NMR signals of SiMe3 and Cp* methyl groups observed with increasing temperature.

 Please wait while we load your content...
Please wait while we load your content...