Graphite oxide–iron oxide nanocomposites as a new class of catalyst for the thermal decomposition of ammonium perchlorate
Abstract
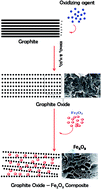
Graphite oxide (GO) is receiving increased attention due to its special surface properties and layered structure for the synthesis of GO containing nanocomposites. It is possible that integration of GO sheets and iron oxide nanoparticles may result in enhanced properties and enlarge the application range. Herein, we report the effect of Fe2O3–GO nanocomposite as a new class of catalyst for the decomposition of ammonium perchlorate (AP), a rocket propellant oxidizer, and study the effect of Fe2O3 : GO ratio on the catalytic activity. The material was characterized by X-ray diffraction and Raman spectroscopy and the formation of Fe2O3 and GO were confirmed. FESEM analysis showed that the Fe2O3 nanoparticles are highly dispersed between and on the graphene layers. With the addition of 3% of the composite with 1 : 1 Fe2O3–GO ratio, the decomposition temperature of AP was reduced by 45 °C, showing a high catalytic activity for the new composite. The high catalytic activity of the in situ synthesized Fe2O3–GO composite may be attributed to the uniform distribution of iron oxide nanoparticles which in turn provide a number of active sites on the surface due to the presence of GO.

 Please wait while we load your content...
Please wait while we load your content...