Removal of anionic azo dye from water with activated graphene oxide: kinetic, equilibrium and thermodynamic modeling
Abstract
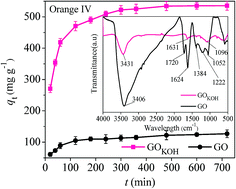
In order to improve the specific surface area value and adsorption capacity of graphene oxide (GO), activated GO (GOKOH) was successfully prepared by high temperature solid state activation with potassium hydroxide, and was used as the adsorbent for the removal of the anionic dye orange IV from water. The obtained adsorbent was characterized by XRD, FTIR, zeta potential, BET and SEM, respectively. The results show that GOKOH had an extraordinary specific surface area of 672.5 m2 g−1 and large adsorption capacity of 606.1 mg g−1 for anionic orange IV, which were much higher than those of GO synthesized by the Hummers method. The activation method can remarkably reduce the amount of electronegative charges on the surface of GO and promote the interaction between GO molecules and anionic dye molecules. The adsorption process was endothermic and spontaneous in nature, following the pseudo-second-order kinetics and the Langmuir isotherm models. The present adsorption studies of anionic dyes revealed the potential of GOKOH to be utilized as adsorbents with super adsorption capacity for water purification.

 Please wait while we load your content...
Please wait while we load your content...