Fabricating a g-C3N4/CuOx heterostructure with tunable valence transition for enhanced photocatalytic activity†
Abstract
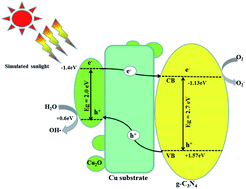
Heterostructured g-C3N4/CuOx nanocomposites were successfully prepared with varying the content of g-C3N4 via a mixed solvent-thermal method. Organic ammonia ethanolamine was used as the reductant to adjust the valence of CuOx. Meanwhile, the bulk g-C3N4 was efficiently exfoliated into ultrathin nanoplates during the synthesis process. It is found that the different contents of g-C3N4 can effectively accelerate the valence transition of Cu in the nanocrystals. That is, the successive appearance of CuO and Cu2O with the enhancement of g-C3N4 content. These heterostructured composites were characterized for structural, morphological, elemental distribution, optical properties, specific surface area and photo-generated carrier separation efficiency by X-ray diffraction (XRD), scanning electron microscopy (SEM), high resolution transmission electron microscopy (HRTEM), Brunauer–Emmett–Teller (BET), diffuse reflectance spectroscopy (DRS) and photoluminescence (PL) spectrum. The photocatalytic performance investigations indicate that the CN5–CuOx is stable enough and shows a remarkable photocatalytic efficiency for degrading methyl orange (MO) under simulated sunlight irradiation, compared with the pure CuOx nanocrystals. The enhanced photocatalytic performance can be attributed to the enlarged surface area, beneficial heterojunction among Cu2O@g-C3N4 and efficient carrier separation efficiency. The abundant Cu substrate in the CuOx nanocrystals functions as a rapid electron/hole pair transfer avenue and inhibits their recombination. Thus the photocatalytic efficiency is tremendously enhanced.

 Please wait while we load your content...
Please wait while we load your content...