Enhancement of the crosslink density, glass transition temperature, and strength of epoxy resin by using functionalized graphene oxide co-curing agents†
Abstract
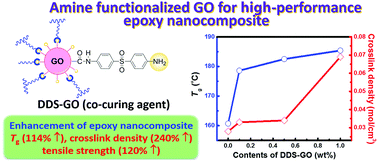
We synthesized diamine-functionalized graphene oxide, DDS–GO and HMDA–GO, by introducing 4,4′-diaminodiphenyl sulfone (DDS) or hexamethylenediamine (HMDA) into the carboxylic acid groups on graphene oxide (GO) via amide bonds. The introduction of diamines was confirmed by analytical methods such as FT-IR, TG-DTA, XPS, AFM, and optical microscopy. Then, we applied DDS–GO and HMDA–GO as co-curing agents for epoxy (EP) nanocomposites that were prepared by mixing bisphenol-A type EP and DDS curing agent (ca. 21 wt%). Interestingly, when 1.0 wt% of DDS–GO was added to the EP/DDS mixture, the crosslink density (CD) increased from 0.028 to 0.069 mol cm−3. Due to the higher CD, both the glass transition temperature and tensile strength of the EP/DDS/DDS–GO nanocomposite effectively improved from 160.7 °C to 183.4 °C and from 87.4 MPa to 110.3 MPa, respectively.
- This article is part of the themed collection: Polymer Chemistry 15th anniversary regional spotlight collection: Asia-Pacific

 Please wait while we load your content...
Please wait while we load your content...