Effects of a trans- or cis-cyclohexane unit on the thermal and rheological properties of semi-aromatic polyamides†
Abstract
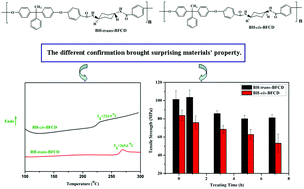
Semi-aromatic monomers containing a cyclohexane unit, prepared using a facile interfacial reaction method from 1,4-cyclohexanediamine (trans- or cis-) and 4-fluorobenzoyl chloride (4-FBC) at room temperature, were reacted with 1,1-bis(4-hydroxyphenyl)-1-phenylethane (BHPPE) to afford two kinds of semi-aromatic polyamide (BH-trans-BFCD and BH-cis-BFCD) in this work. An investigation of thermal properties confirmed high glass transition temperatures (Tg) of 224–265 °C and good thermal stability with initial degradation temperatures (Td) of 445 to 450 °C for the synthesized semi-aromatic polyamides, which were determined by differential scanning calorimetry (DSC) and thermogravimetric analysis (TGA) characterization. It was found that the properties (thermal and mechanical) of BH-trans-BFCD, containing a trans-conformation, were much better than those of BH-cis-BFCD, containing a cis-conformation. Comparing the thermal stability of the resultant semi-aromatic polyamides with a commercial product, PA6T-Dupont, the prepared polyamides treated at 220 °C for 7 h displayed a slightly reduced tensile strength as well as a smaller change in color. According to rheological testing, they were found to have better melt flowability (complex viscosities ranged from 210 to 3070 Pa s at 310 °C to 360 °C). Additionally, their thermal degradation kinetics and the thermal pyrolysis mechanism of these semi-aromatic polyamides were investigated by thermogravimetric analysis and Pyrolysis/Gas Chromatography/Mass (Py-GC/MS) analysis, which further indicated their higher thermal stability and processability.

 Please wait while we load your content...
Please wait while we load your content...