Synthetic analogs of stryphnusin isolated from the marine sponge Stryphnus fortis inhibit acetylcholinesterase with no effect on muscle function or neuromuscular transmission†
Abstract
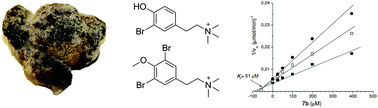
The marine secondary metabolite stryphnusin (1) was isolated from the boreal sponge Stryphnus fortis, collected off the Norwegian coast. Given its resemblance to other natural acetylcholinesterase antagonists, it was evaluated against electric eel acetylcholinesterase and displayed inhibitory activity. A library of twelve synthetic phenethylamine analogs, 2a–7a and 2b–7b, containing tertiary and quaternary amines respectively were synthesized to investigate the individual structural contributions to the activity. Compound 7b was the strongest competitive inhibitor of both acetylcholinesterase and butyrylcholinesterase with IC50 values of 57 and 20 μM, respectively. This inhibitory activity is one order of magnitude higher than the positive control physostigmine, and is comparable with several other marine acetylcholinesterase inhibitors. The physiological effect of compound 7b on muscle function and neuromuscular transmission was studied and revealed a selective mode of action at the investigated concentration. This data is of importance as the interference of therapeutic acetylcholinesterase inhibitors with neuromuscular transmission can be problematic and lead to unwanted side effects. The current findings also provide additional insights into the structure–activity relationship of both natural and synthetic acetylcholinesterase inhibitors.

 Please wait while we load your content...
Please wait while we load your content...