A new NBS/oxone promoted one pot cascade synthesis of 2-aminobenzimidazoles/2-aminobenzoxazoles: a facile approach†
Abstract
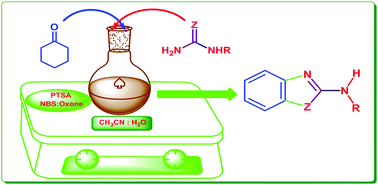
A new strategy for the synthesis of 2-aminobenzimidazoles via a reaction of α-halogenated cyclohexanone with guanidine is reported. It provides a facile N-bromosuccinimide (NBS) mediated transition metal free, sustainable path for the synthesis of 2-substituted azoles. NBS along with the oxone promotes in situ halogenation of cyclohexanone, which in turn, when allowed to react with guanidine, afforded 2-aminobenzimidazoles (2-ABI). This eco-friendly protocol provides a competent laboratory synthesis method associated with significant advantages such as greater selectivity, cost-efficiency, mild reaction conditions, absence of chromatographic purification, clean reaction profile, and a simple work-up procedure to furnish high yields. The structures of all the synthesized compounds were confirmed on the basis of IR, 1H and 13C NMR, mass spectrometry, and elemental analyses data. In addition, we have also extended the scope of this strategy for obtaining 2-amino substituted benzoxazoles.

 Please wait while we load your content...
Please wait while we load your content...