Two-dimensional coordination polymers and metal–organic gels of symmetrical and unsymmetrical dipyridyl β-diketones: luminescence, dye absorption and mechanical properties†
Abstract
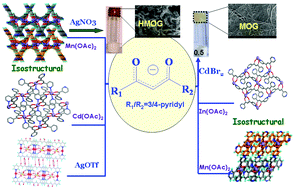
The ability of HL1 (1,3-dipyridin-3-yl-propane-1,3-dione) and HL2 (1-pyridin-4-yl-3pyridin-3-yl-propane-1,3-dione) to form coordination polymers and metal–organic gels with Mn(II)/Cd(II)/Zn(II)/Ag(I) was explored. The reactions of HL1 with acetate salts of Mn(II), Cd(II) and Zn(II) were found to yield coordination polymers 1–3, respectively, with a common formula of [M(L1)2]n. The crystal structure analyses revealed that complexes 1 and 2 are isostructural with a 2D corrugated layer in which M(L1)2 building blocks are interconnected to form M4(L1)4 macrocycles with square geometries. Although the complex 3 contains a similar layer as the above two, it differs significantly due to the differences in the geometry of L1. The 2D-layers interdigitate through aromatic interactions in 1 and 2, whereas the extent of such interdigitation is less in 3. The unsymmetrical HL2 was also found to form coordination polymer [Mn(L2)2]n, 4, upon treatment with Mn(OAc)2. The complex 4 exhibits a similar 2D-network as in 3 due to the fact that only 3-pyridyl is involved in coordination leaving 4-pyridyl uncoordinated. Further, the reaction of complex 1 with AgNO3 in DMSO was found to form a hetero-metal–organic gel (HMOG) while the reaction of HL1 with CdBr2 resulted in the formation of a metal–organic gel (MOG). The gel materials were characterized by various microscopic and rheological studies. Further, the presence of both Mn(II) and Ag(I) in HMOG was confirmed by EDAX experiments. Both the gels were found to exhibit thixotropic behaviour, and the firmness of MOG was found to be much higher than that of HMOG. The reaction of Ag(I) salt with HL1 resulted in single crystals of the complex [Ag(HL1)(O3SCF3)]n, 5, which gave significant insights into the structures of gels. The luminescence studies indicate that complexes 2, 3 and MOG exhibit better luminescence compared to 1 and HMOG. Similarly, the MOG was found to be a better dye absorbing material than HMOG.

 Please wait while we load your content...
Please wait while we load your content...