Waterborne polyurethanes from CO2 based polyols with comprehensive hydrolysis/oxidation resistance†
Abstract
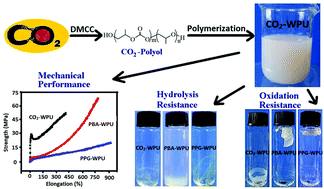
Carbon dioxide based oligo(carbonate–ether) diols (CO2-polyols) with both carbonate units and ether units in one polymer chain were prepared by copolymerization of CO2 and propylene oxide (PO) using a zinc–cobalt double metal complex as the catalyst, and used to prepare CO2 based waterborne polyurethanes (CO2-WPUs). The carbonate units in CO2-polyols improved the mechanical and oxidation resistance properties of CO2-WPUs, while the ether units in CO2-polyols enhanced the hydrolysis resistance of CO2-WPUs. The tensile strength of CO2-WPUs didn't show any obvious drop during immersion in a 0.25% sodium hydroxide solution, whereas that of oligoesterol based WPUs dropped over 50% after 300 min, and lost their mechanical properties after 520 min of immersion. Meanwhile, the retention of the tensile strength of the CO2-WPUs was ca. 72% even after 46 h of immersion in a 6 wt% H2O2 solution, while it was only ca. 32% for the oligoetherol based WPUs. Moreover, the thermal–mechanical performance of CO2-WPU films can be conveniently improved by adjusting the carbonate unit content (CU%) in CO2-polyols, i.e. when CU% in CO2-polyol increased from 30% to 66%, the glass transition temperature (Tg) increased from −7.8 °C to 18.8 °C, accompanied by an increase of tensile strength from 35.6 MPa to 52.2 MPa, and a decrease of elongation at break from 630% to 410%. This work suggests that the CO2-WPU may be a promising alternative to conventional WPU whose oligoetherol and oligoesterol were from fossil resources, and its comprehensive hydrolysis/oxidation resistance may be a bonus unavailable from common oligomerol based WPUs.

 Please wait while we load your content...
Please wait while we load your content...