MIL-101 promotes the efficient aerobic oxidative desulfurization of dibenzothiophenes†
Abstract
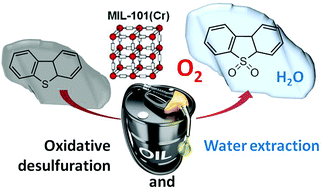
MIL-101 promotes aerobic oxidation in n-dodecane of dibenzothiophene (DBT) and its methyl-substituted derivatives to their corresponding sulfones with complete selectivity, without observation of the sulfoxide. DBT sulfones can be completely separated from n-dodecane by water extraction. MIL-101(Cr) without the need of pre-activation was found to be more convenient than the also-active MIL-101(Fe) analog. The reaction exhibits an induction period due to the diffusion inside the pore system of the solvent or oxygen and it is not observed if the MIL-101 sample is first in contact with the solvent at the reaction temperature for a sufficiently long time. MIL-101 is reusable for at least five times without any sign of deactivation according to the time-conversion plots. Evidence by electron paramagnetic resonance spectroscopy detecting the hydroperoxide radical adduct with a spin trapping agent and Raman spectroscopy detection of superoxide supports that the process is an auto-oxidation reaction initiated by MIL-101 following the expected radical chain mechanism inside the MIL-101 cages.

 Please wait while we load your content...
Please wait while we load your content...