Well-defined coinage metal transfer agents for the synthesis of NHC-based nickel, rhodium and palladium macrocycles†
Abstract
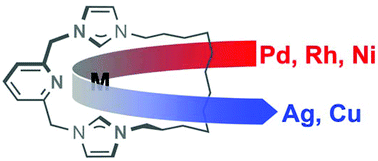
With a view to use as carbene transfer agents, well-defined silver(I) and copper(I) complexes of a macrocyclic NHC-based pincer ligand, bearing a central lutidine donor and a dodecamethylene spacer [CNC–(CH2)12, 1], have been prepared. Although the silver adduct is characterised by X-ray diffraction as a dinuclear species anti-[Ag(μ-1)]22+, variable temperature measurements indicate dynamic structural interchange in solution involving fragmentation into mononuclear [Ag(1)]+ on the NMR time scale. In contrast, a mononuclear structure is evident in both solution and the solid-state for the analogous copper adduct partnered with the weakly coordinating [BArF4]− counter anion. A related copper derivative, bearing instead the more coordinating cuprous bromide dianion [Cu2Br4]2−, is notable for the adoption of an interesting tetranuclear assembly in the solid-state, featuring two cuprophilic interactions and two bridging NHC donors, but is not retained on dissolution. Coinage metal precursors [M(1)]n[BArF4]n (M = Ag, n = 2; M = Cu, n = 1) both act as carbene transfer agents to afford palladium, rhodium and nickel complexes of 1 and the effectiveness of these precursors has been evaluated under equivalent reaction conditions.


 Please wait while we load your content...
Please wait while we load your content...