Phosphaalkene-oxazoline copolymers with styrene as chiral ligands for rhodium(i)†
Abstract
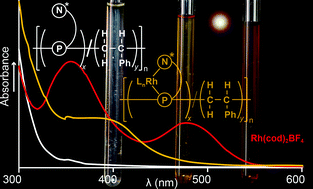
The radical-initiated copolymerization of phosphaalkene-oxazoline, MesP![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C(Ph)CMe2Ox [1, Ox = CNOCH(iPr)CH2] with different loadings of styrene affords poly(methylenephosphine-co-styrene)s [2a (1 : S = 1 : 2): Mw = 7400 g mol−1, PDI = 1.1; 2b (1 : S = 1 : 5): Mw = 18 000 g mol−1, PDI = 1.2; 2c (1 : S = 1 : 10): Mw = 16 000 g mol−1, PDI = 1.3]. Copolymers 2a–2c are demonstrated as viable macromolecular ligands for rhodium(I). By comparison with the crystallographically characterized model P,N-bidentate complex, [Mes(Me)P–CH(Ph)CMe2Ox·Rh(cod)]BF4, the polymer complexes [2·Rh(cod)]BF4 were prepared. The macromolecular metal complexes were characterized by GPC {for [2a·Rh(cod)]BF4: Mw = 14 000 g mol−1, PDI = 1.2}, UV/Vis spectroscopy, 1H, 13C and 31P NMR spectroscopy. Integration of the 31P NMR spectra of mixtures of 2 and [Rh(cod)2]BF4 permitted the determination of the mol% of incorporation of monomer 1 in copolymer 2 (2a: 17%; 2b: 5%; 2c: 4%). These results compared favorably with those determined by elemental analysis (2a: 17%; 2b: 6%).
C(Ph)CMe2Ox [1, Ox = CNOCH(iPr)CH2] with different loadings of styrene affords poly(methylenephosphine-co-styrene)s [2a (1 : S = 1 : 2): Mw = 7400 g mol−1, PDI = 1.1; 2b (1 : S = 1 : 5): Mw = 18 000 g mol−1, PDI = 1.2; 2c (1 : S = 1 : 10): Mw = 16 000 g mol−1, PDI = 1.3]. Copolymers 2a–2c are demonstrated as viable macromolecular ligands for rhodium(I). By comparison with the crystallographically characterized model P,N-bidentate complex, [Mes(Me)P–CH(Ph)CMe2Ox·Rh(cod)]BF4, the polymer complexes [2·Rh(cod)]BF4 were prepared. The macromolecular metal complexes were characterized by GPC {for [2a·Rh(cod)]BF4: Mw = 14 000 g mol−1, PDI = 1.2}, UV/Vis spectroscopy, 1H, 13C and 31P NMR spectroscopy. Integration of the 31P NMR spectra of mixtures of 2 and [Rh(cod)2]BF4 permitted the determination of the mol% of incorporation of monomer 1 in copolymer 2 (2a: 17%; 2b: 5%; 2c: 4%). These results compared favorably with those determined by elemental analysis (2a: 17%; 2b: 6%).

 Please wait while we load your content...
Please wait while we load your content...