The substituent effect of 2-R-o-carborane on the photophysical properties of iridium(iii) cyclometalates†
Abstract
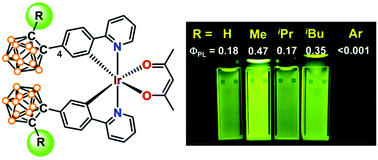
A family of heteroleptic iridium(III) cyclometalates, [4-(2-RCB)ppy]2Ir(acac) (3c–3g; CB = o-carboran-1-yl; ppy = 2-phenylpyridinato-C2,N; acac = acetylacetonate; R = iPr (3c), iBu (3d), Ph (3e), CF3C6H4 (3f), C6F5 (3g)) with various 2-R substituted o-carboranes at the 4-position in the phenyl ring of the ppy ligand, were prepared. The X-ray crystal structure of all complexes, including 3a (R = H) and 3b (R = Me), showed that while the carboranyl C–C bond distance increases with the increasing steric effect of the 2-R substituent (3a–3d), the bond distance is more likely to be influenced by the electronic effect of the substituent for the 2-aryl substituted complexes (3e–3g). The absorption and emission bands of all complexes were red-shifted with respect to those of the parent (ppy)2Ir(acac) (4). While 3a–3d exhibited intense green phosphorescence with good quantum efficiency in toluene (ΦPL = 0.17–0.47), the complexes were poorly emissive in THF (ΦPL < 0.004). In a PMMA film, however, 3a–3d became substantially emissive (ΦPL = 0.11–0.14, 8 wt% Ir), albeit less emissive than 4 (ΦPL = 0.15). In contrast, the 2-aryl substituted 3e–3g were almost non-emissive in both the solution and solid states. All complexes underwent facile carborane-centered, electrochemical reduction (Eonset = −2.01 to −1.22 V). The potential values of these reductions increased with increases in the electron accepting ability of the 2-R group (H < alkyl < aryl) and were largely shifted anodically with respect to those of 4 and 5-carborane substituted complexes. This reduction behavior of 3a–3g implies that the LUMO contribution of 2-R-o-carborane units increases through the substitution at the 4-position of the ppy ligand and is influenced by the nature of the 2-R group, which may be responsible for facile carboranyl C–C bond variation leading to efficient quenching of the emissive excited states.


 Please wait while we load your content...
Please wait while we load your content...