Insight into the mechanism and site-selectivity of Rh2II,II(esp)2-catalyzed intermolecular C–H amination†
Abstract
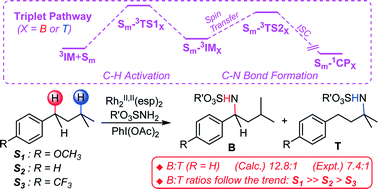
The mechanisms and site-selective determinants of Rh2II,II(esp)2-catalyzed intermolecular C–H bond aminations of three isoamylbenzene-derived substrates, p-R–C6H4(CH2)2CH(CH3)2 (R = OCH3, H, CF3), have been investigated by the density functional theory (BPW91) method. Our calculations show that both σ-type and π-type 3c/4e bonds exist in dirhodium nitrene Rh2(esp)2–NSO3CH2CF3, and both the LUMO and SOMO2 of Rh2-nitrene comprise a set of three-center (Rh–Rh–N) π* orbitals. The three-center nature of these orbitals results in the nitrenoid nitrogen atom being of strong electrophilic reactivity, thus greatly facilitating C–H amination. A triplet stepwise pathway is favorable for Rh2II,II(esp)2-catalyzed intermolecular C–H amination. Calculation results show that both electronic and steric effects are the principal determinants of the benzylic-to-tertiary ratio for intermolecular C–H amination of S2. A theoretical examination of how para substituent R (R = OCH3, H, CF3) affects site-selectivity is also highlighted. The calculated isoamylbenzene benzylic-to-tertiary C–H amination ratio (B : T) is 12.8 : 1, which is in good agreement with the experimental value (B : T = 7.4 : 1). The para substituents impact the reactivity of the benzylic rather than the tertiary C–H bond by electronic effects. The strongly electron-donating substituent reduces the activation barrier of benzylic C–H amination via a p–π conjugation, thus the B : T product ratio is increased. The calculated B : T ratios of p-methoxy-isoamyl-benzene (S1), isoamylbenzene (S2) and p-trifluoromethyl-isoamylbenzene (S3) follow the following trend: S1 ≫ S2 > S3, which is in accordance with the experimental result reported by J. Du Bois’ group.

 Please wait while we load your content...
Please wait while we load your content...