An investigation on the selective hydrodealkylation of C9+ aromatics over alkali-treated Pt/H-ZSM-5 zeolites
Abstract
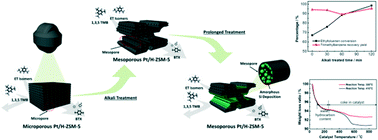
Among heterogeneous catalysts, Pt/H-ZSM-5 zeolite has been widely utilized as an acid catalyst for reforming hydrocarbons, which converts heavy reformate into value-added BTX. In this work, we demonstrate the effect of the secondary pore structure in Pt/H-ZSM-5 catalysts on the catalytic performance for selective hydrodealkylation of C9+ aromatics. Mesoporous Pt/H-ZSM-5 catalyst with a bimodal pore system is simply synthesized by caustic treatment with sodium hydroxide solution. Multiple techniques, such as SEM, TEM, ICP, and N2 adsorption techniques, are employed for the physical characterization of conventional Pt/H-ZSM-5 and mesoporous Pt/H-ZSM-5. The as-prepared Pt/H-ZSM-5 catalysts are investigated for their catalytic performance as a function of alkali-treatment time, including ET (ethyl toluene) conversion and TMB (trimethyl benzene) recovery yield. The alkali-treated Pt/H-ZSM-5 for 120 min (Pt/H-ZSM-5(120)) demonstrated excellent catalytic activities on the selective hydrodealkylation of C9+ aromatics, even at a low reaction temperature. The exceptional catalytic behavior at low temperature can be attributed to the generation of secondary mesoporous systems within the zeolite catalysts, which can promote the mass transfer of reactant and generated products and effectively suppress coke deposition. The findings reported herein suggest that this strategy can be effectively used not only to reduce energy consumption but also to provide a cost-saving process for producing BTX.

 Please wait while we load your content...
Please wait while we load your content...