Amount of tungsten dopant influencing the photocatalytic water oxidation activity of LaTiO2N crystals grown directly by an NH3-assisted flux method
Abstract
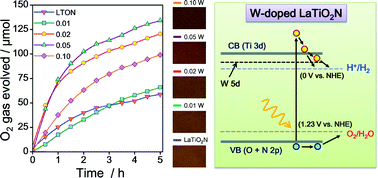
Solar water splitting technologies for hydrogen generation using visible-light-active (oxy)nitride photocatalysts have been intensively studied to achieve a greater supply of clean and renewable energy. Here, we have investigated the amount of tungsten dopant influencing the photocatalytic water oxidation activity of LaTiO2N (LTON) crystals grown directly by an NH3-assisted KCl flux method in the absence of a cocatalyst. With the maximum amount of tungsten dopant (x = 0.10), LaWO0.6N2.4 was formed as a minor phase to a W-doped LaTiO2N phase (W-LTON). The optimum W-doped LaTiO2N crystals were grown with the tungsten amount of x = 0.05, showing a nearly three-fold higher O2 evolution rate (51 μmol h−1) compared to pure LaTiO2N crystals (18 μmol h–1). The improvement in the O2 evolution rate of the LaTiO2N crystals was presumably due to the improved conductivity, enhanced light absorption and efficient charge separation stemmed from tungsten doping. According to first-principles density function theory (DFT) calculations, although no significant change in band gap by tungsten doping was observed, slight broadening of valence and conduction bands was noted in W-LTON which may improve the electron/hole conductivity due to reduction of the effective mass of electrons. Also, tungsten doping was beneficial in creating effective defects and in enhancing the photocatalytic water oxidation activity of the LaTiO2N crystals.


 Please wait while we load your content...
Please wait while we load your content...