Ligand ordering determines the catalytic response of hybrid palladium nanoparticles in hydrogenation†
Abstract
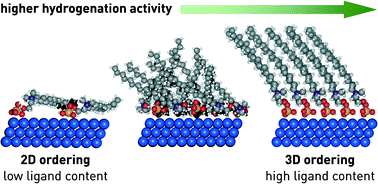
Supported palladium nanoparticles, prepared by reducing the active metal in the presence of the hexadecyl(2-hydroxyethyl)dimethylammonium dihydrogen-phosphate (HHDMA) ligand and depositing the resulting colloids on titanium silicate (TiSi2O6), represent a proven alternative to the archetypal poisoned catalysts in industrially-relevant selective hydrogenations. To date, a key aspect in the design of these hybrid nanocatalysts remains unaddressed, namely the impact of the ligand content on the catalytic behaviour. In order to assess the structural and associated catalytic implications of this variable, we have prepared a series of Pd-HHDMA/TiSi2O6 catalysts with different HHDMA content (0.3–16.8 wt%), keeping the average particle size (5 nm) and Pd content (0.3 wt%) constant. The materials are characterised with a toolbox of methods, including advanced microscopy and solid-state nuclear magnetic resonance, in order to assess the structure metal–ligand interface and the mobility of the alkyl chain. Continuous-flow three-phase hydrogenations of short-chain acetylenic compounds, nitriles, and carbonyls reveal an increase in the catalytic activity with the ligand content. Density Functional Theory indicates that the ligand behaves as a self-assembled monolayer, changing its adsorption configuration as a function of the HHDMA concentration. At low coverage, the organic layer lies almost flat on the surface of the metal nanoparticle blocking a large number of metal sites and resembling a two-dimensional catalyst; high HHDMA coverages favour an extended three-dimensional configuration of the alkyl chain, and consequently a lower fraction of Pd sites are poisoned. These results provide new fundamental insights into the role of the ligand on the catalytic activity and can enable the design of hybrid nanocatalysts with optimised performance.

 Please wait while we load your content...
Please wait while we load your content...