Mass spectrometric analysis of residual clenbuterol enantiomers in swine, beef and lamb meat by liquid chromatography tandem mass spectrometry
Abstract
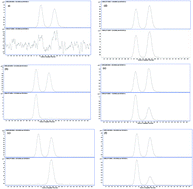
A method for determining the ratio of clenbuterol enantiomers (R/S ratio) in edible meat by high performance liquid chromatography-mass spectrometry was developed and validated, and clenbuterol chiral determination in beef and lamb meat were first reported. The practical procedure involves acid extraction followed by one solid-phase clean-up step with weak cation-exchange resins. 2H9-(+/−)-clenbuterol was used as an internal standard. R-(−)-Clenbuterol and S-(+)-clenbuterol were completely baseline separated and detected by HPLC-MS/MS. The limit of detection (LOD) for the enantiomers of clenbuterol was 0.1 μg kg−1 and the limit of quantification (LOQ) was 0.2 μg kg−1. The spiked R-(−)-clenbuterol or S-(+)-clenbuterol in the blank sample was stable and the conversion between R-(−)-clenbuterol and S-(+)-clenbuterol was not detected in the sample procedure. The R/S ratio of racemic clenbuterol spiked in the blank sample ranged between 0.92 and 1.06 at high and middle and the average value of LOQ concentration was 1.00. The R/S ratio in a total of 94 clenbuterol residues in edible meat samples collected from the market was calculated using the current method, and the R/S ratio in swine meat (5 samples) ranged from 0.65 to 0.77; the R/S ratio in beef meat (73 samples) ranged from 0.89 to 2.42; and the R/S ratio in lamb meat (16 samples) ranged from 1.45 to 2.82. This is the first time that the different R/S ratios in farm animal meat have been calculated and reported. The effects of the R/S ratio differences in food safety, bioactivity and doping control test are still unknown but is an area of future investigation. This technique can be considered as a starting point for the distribution and pharmacokinetics of clenbuterol enantiomers in livestock.

 Please wait while we load your content...
Please wait while we load your content...