Alkali-stable and highly anion conducting poly(phenylene oxide)s carrying quaternary piperidinium cations†
Abstract
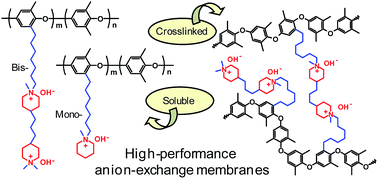
New durable and hydroxide ion conducting anion-exchange membranes (AEMs) are currently required in order to develop alkaline fuel cells into efficient and clean energy conversion devices. In the present work we have attached quaternary piperidinium (QPi) cations to poly(2,6-dimethyl-1,4-phenylene oxide)s (PPOs) via flexible alkyl spacer chains with the aim to prepare AEMs. The bromine atoms of bromoalkylated PPOs were displaced in Menshutkin reactions to attach one or two QPi groups, respectively, via heptyl spacers. The cationic polymers have excellent solubility in, e.g., methanol, dimethylsulfoxide and N-methyl-2-pyrrolidone at room temperature, and form tough and transparent membranes. AEMs with bis-QPi side chains efficiently form ionic clusters and reach very high hydroxide ion conductivities, up to 69 and 186 mS cm−1 at 20 and 80 °C, respectively. The AEMs further have excellent alkaline stability, and 1H NMR analysis showed no degradation of the AEMs after storage in 1 M NaOH at 90 °C during 8 days. Thermogravimetry indicated decomposition only above 225 °C. The AEM properties were further tuned by controlled formation of bis-QPi crosslinks through an efficient reaction between bromoalkylated PPO and 4,4′-trimethylenebis(1-methylpiperidine) during a reactive membrane casting process. In conclusion, alkali-stable and highly conductive AEMs can be prepared by placing cycloaliphatic quaternary ammonium cations on flexible side chains and crosslinks.

 Please wait while we load your content...
Please wait while we load your content...