Preparation of oxocene terpenes. The first enantiospecific synthesis of cytotoxic arenaran A†
Abstract
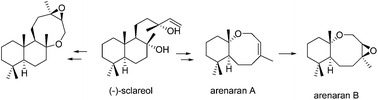
The first syntheses of cytotoxic marine arenarans A and B starting from commercial (−)-sclareol are reported. The oxocene ring of the target compound is formed via ring-closing metathesis, a process that depends on certain structural requirements. The trans-fused structure of the natural product is confirmed by comparison with the cis-fused isomer, which was synthesized. This synthetic strategy is also applicable to the synthesis of other oxocene terpenes.

 Please wait while we load your content...
Please wait while we load your content...