Complexity in structure-directed prebiotic chemistry. Unexpected compositional richness from competing reactants in tetrapyrrole formation†
Abstract
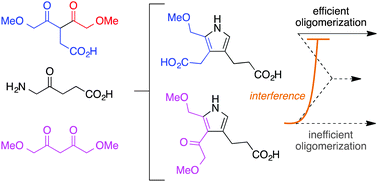
Robustness is a key feature of any credible prebiotic reaction process. Two studies are presented concerning the role of analogous yet defective reactants (spanning a range of reactivity) in a model for the prebiogenesis of tetrapyrrole macrocycles in aqueous solution under mild conditions. The reaction entails condensation of an α-aminoketone (e.g., δ-aminolevulinic acid) with a 1,5-dimethoxypentan-2,4-dione bearing a 3-acetic acid unit (β-diketone 1-AcOH) to form a porphobilinogen analogue, which self-condenses to form a mixture of porphyrinogens. Here, the inclusion of an analogous β-diketone lacking the acetic acid substituent (1,5-dimethoxypentan-2,4-dione, 1-H) formed a partially defective (weakly reactive) pyrrole, which upon combinatorial reaction with the normal pyrrole (derived from 1-AcOH) severely crimped the yield of the latter yet all porphyrinogens derivable from both β-diketones 1-H and 1-AcOH were observed upon mass spectrometric analysis. Detection of the porphyrinogens by mass spectrometry revealed a compositional richness beneath the limits of detection by routine absorption spectroscopy. A similar reaction of 1-amino-2-butanone and 1,5-dimethoxy-3-methylpentan-2,4-dione in aqueous micelles leading to etioporphyrinogens was examined in the presence of four β-diketones. Linear offsets of yield as well as combinatorial inhibition were observed depending on the dione structure. Taken together, the results provide a nuanced, quantitative view of the effects of reactant analogues in a chemical model of a structure-directed prebiotic process. The adverse effects of such reactant analogues provide a cautionary tale about the viability of structure-directed processes as a sole means for creation of architecturally complex molecules in a primordial broth.

 Please wait while we load your content...
Please wait while we load your content...