Complexity in structure-directed prebiotic chemistry. Reaction bifurcation from a β-diketone in tetrapyrrole formation
Abstract
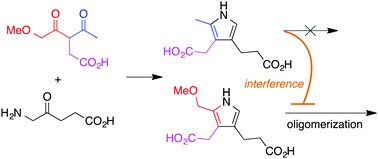
An early stage of prebiotic chemistry is regarded to have entailed structure-directed reactions, where the products depend on intrinsic properties of the reactants without guidance by any enzyme-like catalysts. A chemical model for the prebiogenesis of tetrapyrrole macrocycles entails condensation of an α-aminoketone (e.g., δ-aminolevulinic acid, ALA) with a 1,5-dimethoxypentan-2,4-dione bearing a 3-alkyl substituent (β-diketone, hereafter referred to as a dione) to form a porphobilinogen analogue, which self-condenses to form a porphyrinogen. The reaction proceeds under mild conditions (60 °C, pH 7, 24 h). Here, the reaction of a mono-methoxy substituted dione analogue with ALA formed two pyrroles, a normal pyrrole equipped for oligomerization to form uroporphyrinogens and a defective pyrrole that cannot undergo oligomerization. The defective pyrrole has one open α-position and one α-methyl group, and as such, can terminate the oligomerization of the normal pyrrole, thereby thwarting reaction leading to the porphyrinogen. The small defect of a single missing methoxy group has outside adverse effects in a complex reaction process. As a case study of relevance to prebiotic chemistry, the results highlight limitations of structure-directed processes.

 Please wait while we load your content...
Please wait while we load your content...