Selective hydrodeoxygenation of lignin-derived phenols to alkyl cyclohexanols over a Ru-solid base bifunctional catalyst†
Abstract
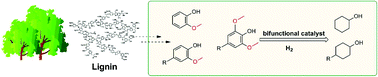
Cyclohexanol and alkyl cyclohexanol are important chemical intermediates. It is meaningful to prepare cyclohexanols from non-fossil-based biomass. Here we report Ru/ZrO2–La(OH)3, a metal-solid base bifunctional catalyst, to show its excellent performance on the partial hydrodeoxygenation of lignin-derived phenols. Guaiacol could be converted to cyclohexanol with a 91.6% yield in water. Alkyl phenols with one or two methoxy groups were converted into alkyl cyclohexanols with yields over 86.9%. The catalyst had good activity of removing a methoxy group and retaining a hydroxyl group. In this catalyst, Zr and La interacted with each other to form a mixed (hydr)oxide, thus making ZrO2–La(OH)3 a stable support. Ru was highly dispersed on the ZrLa support. The pathway from guaiacol to cyclohexanol was investigated and proposed as two parallel ways, demethoxylation followed by hydrogenation (I), the saturation of the aromatic ring through hydrogenation and then demethoxylation through direct hydrogenolysis (II).

 Please wait while we load your content...
Please wait while we load your content...