An indirect generation of 1D MII-2,5-dihydroxybenzoquinone coordination polymers, their structural rearrangements and generation of materials with a high affinity for H2, CO2 and CH4†
Abstract
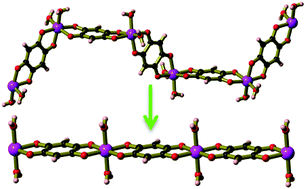
A series of solid-state structural transformations are found to accompany desolvation of relatively simple coordination polymers to yield materials that exhibit unexpected gas sorbing properties. Reaction of 1,2,4,5-tetrahydroxybenzene with MII salts (M = Mg, or Zn) in an alcohol/water solution in the presence of air affords cis-MII(C6H2O4−II)(H2O)2·2H2O·xROH, (M = Mg, or Zn), crankshaft-like chains in which the absolute configurations of the chiral metal centres follow the pattern ⋯Δ Δ Λ Λ Δ Δ Λ Λ⋯, and are hydrogen bonded together to generate spacious channels. When crystals of the crankshaft chain are air dried the crystals undergo a single crystal-to-powder rearrangement to form linear trans-MII(C6H2O4−II)(H2O)2 chains. Further dehydration yields microporous solids that reversibly sorb H2, CH4 and CO2 with high sorption enthalpies.


 Please wait while we load your content...
Please wait while we load your content...