Alloying ZnS in the hexagonal phase to create high-performing transparent conducting materials†
Abstract
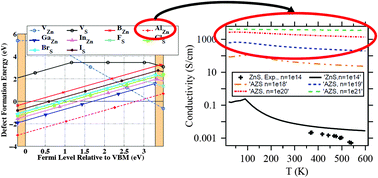
Alloyed zinc sulfide (ZnS) has shown promise as a relatively inexpensive and earth-abundant transparent conducting material (TCM). Though Cu-doped ZnS has been identified as a high-performing p-type TCM, the corresponding n-doped ZnS has, to date, been challenging to synthesize in a controlled manner; this is because the dopant atoms compete with hole-inducing zinc vacancies near the conduction band minimum as the most thermodynamically stable intrinsic point defects. We thus aim to identify the most promising n-type ZnS-based TCM, with the optimal combination of physical stability, transparency, and electrical conductivity. Using a relatively new method for calculating the free energy of both the sphalerite (cubic) and wurtzite (hexagonal) phases of undoped and doped ZnS, we find that doped ZnS is more stable in the hexagonal structure. This, for the first time, fundamentally explains previous experimental observations of the coexistence of both phases in doped ZnS; hence, it profoundly impacts future work on sulfide TCMs. We also employ hybrid density functional theory calculations and a new carrier transport model, AMSET (ab initio model for mobility and Seebeck coefficient using the Boltzmann transport equation), to analyze the defect physics and electron mobility of the different cation- (B, Al, Ga, In) and anion-doped (F, Cl, Br, I) ZnS, in both the cubic and hexagonal phases, at various dopant compositions, temperatures, and carrier concentrations. Among all doped ZnS candidates, Al-doped ZnS (AZS) exhibits the highest dopant solubility, largest electronic band gap, and highest electrical conductivity of 3830, 1905, and 321 S cm−1, corresponding to the possible carrier concentrations of n = 1021, 1020, and 1019 cm−3, respectively, at the optimal 6.25% dopant concentration of Al and the temperature of 300 K.

 Please wait while we load your content...
Please wait while we load your content...