Do poly(epsilon-caprolactone) lipid-core nanocapsules induce oxidative or inflammatory damage after in vivo subchronic treatment?
Abstract
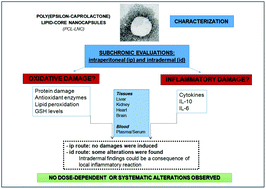
Among the toxicity mechanisms linked to nanoparticles (NPs), oxidative stress (OS) and inflammation are, in general, presumed to mediate their toxicological responses. In a previous toxicological screening, we evaluated whether lipid-core nanocapsules (LNC) induced in vivo alterations, however, no mechanisms of toxicity were determined. The present study aimed to investigate oxidative stress (OS) and inflammatory markers following poly(ε-caprolactone) (PCL) LNC intradermal and intraperitoneal subchronic treatment. OS biomarkers and cytokines were analyzed in blood and/or tissue homogenates. We report that PCL-LNC did not induce lipid peroxidation in plasma, liver, kidney and cardiac tissues, except for the brain after id administration of the highest dose. In contrast, enhanced protein damage by carbonylation was found in the intermediate and highest ip doses and a polysorbate 80 (PS80) group compared to a saline group and also high protein nitrosylation in the highest id dose. In general, no important alterations were found in the activities of the antioxidant enzymes SOD and CAT compared to controls. IL-10 levels were only decreased after the highest id dose and the PS80-group compared to the saline group. Overall, the tested PCL-LNC, especially via ip, did not alter the oxidative status in a systematic repeated-dose approach, thus providing evidence for a safe use of these biodegradable PCL nanocapsules as systemic drug nanocarriers. However, the intradermal results could be a consequence of a local inflammatory reaction which resulted in modified oxidative status and inflammation, requiring further investigation or alternative routes of administration.
- This article is part of the themed collection: Nanotoxicology

 Please wait while we load your content...
Please wait while we load your content...