In situ X-ray diffraction study of the controlled oxidation and reduction in the V–O system for the synthesis of VO2 and V2O3 thin films†
Abstract
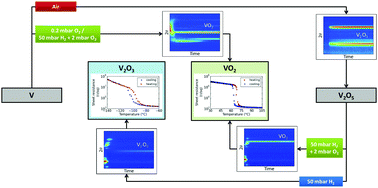
VO2 and V2O3 thin films have been prepared by controlled oxidation and reduction reactions in the vanadium–oxygen system. During these reactions, crystalline phase formation and stability were characterized by means of in situ X-ray diffraction. Oxidation of vanadium thin films was carried out over a wide range of oxygen partial pressures between 0.2 and 200 mbar and temperatures between 430 °C and 615 °C. Depending on the oxygen partial pressures and temperatures, VO2, V6O13 and V2O5 could be obtained as pure or mixed phases. Reduction of V2O5 in 50 mbar H2 resulted in a continuous reduction to V2O3. Stabilization of the VO2 phase was obtained by adding low O2 concentrations in the range from 0.2 to 2 mbar to the H2 gas, a method which proved to be successful also for the controlled oxidation of vanadium to VO2. The semiconductor−metal transition was observed by means of temperature dependent sheet resistance measurements. VO2 films prepared by the oxidation of vanadium at low oxygen partial pressures were characterized by a 3 orders of magnitude decrease in resistance during transition. Annealing in air only yielded comparable switching ratios when the annealing time was carefully optimized. Both the VO2 films prepared by oxidation of vanadium or reduction of V2O5 in the mixture of H2 and O2 showed 4 to 5 orders of magnitude switching, which is close to the best reported values for bulk, single-crystal VO2. This excellent switching performance is believed to originate from a decreased level of defects at grain boundaries and in the bulk. In addition, the V2O3 films prepared by reduction of V2O5 showed a 3 orders of magnitude increase in resistance near −100 °C. Our results provide methods for transforming vanadium oxide phases into VO2 and V2O3 with high resistance switching ratios.

 Please wait while we load your content...
Please wait while we load your content...