La4Ni3O10−δ as an efficient solid oxide fuel cell cathode: electrochemical properties versus microstructure
Abstract
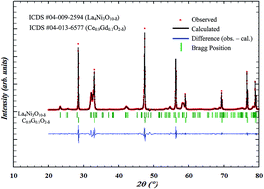
The higher-order Ruddlesden–Popper phase La4Ni3O10−δ is prepared for the first time by electrostatic spray deposition (ESD) on a CGO (Ce0.9Gd0.1O2−δ) electrolyte and evaluated as an intermediate temperature solid oxide fuel cell cathode. Different and innovative microstructures are obtained by varying the deposition time, nozzle to substrate distance, substrate temperature, solution flow rate, concentration and solvents. Single phase La4Ni3O10−δ films crystallize in an orthorhombic layered Ruddlesden–Popper (n = 3) structure after calcination at 950 °C for 8 h in air and is maintained after further sintering at 1100 °C for 6 h in air. The surface morphology, observed by SEM-FEG, is strongly influenced by the solution concentration, the nature of the solvents and the deposition temperature. The electrochemical properties are found to be strongly dependent on the microstructure of the cathode films. The lowest polarization resistance values obtained for the double layer (a 3-D coral-type film by ESD topped by a screen-printed layer) cathode are 2.01, 0.30 and 0.05 Ω cm2 at 600 °C, 700 °C and 800 °C, respectively. To the best of our knowledge, this La4Ni3O10−δ cathode shows the best performance reported to date for this composition.

 Please wait while we load your content...
Please wait while we load your content...