Exceptionally highly performing Na-ion battery anode using crystalline SnO2 nanoparticles confined in mesoporous carbon†
Abstract
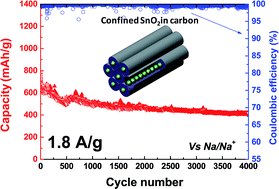
Confined and unconfined SnO2 nanoparticles in the pores of mesoporous carbon were prepared and tested as anode materials vs. Na. Both composites present small crystalline SnO2 particles (∼3 nm) but different location and dispersion in the carbon matrix. When the particles are homogeneously distributed and confined in the carbon pores, an initial reversible capacity of 780 mA h g−1 is achieved with unprecedented capacity retention of 80 and 54% after 100 and 4000 cycles, respectively, at a high current rate (50 C, 1800 mA g−1). Unexpectedly, over two current rate variation cycles from 1 C to 500 C, the composite recovers 81% and 97%, respectively after returning from the 500 C to the 1 C rate. To our knowledge, no other material with such a long cycling life and superior performance in terms of capacity and rate capability has been reported so far for sodium ion batteries. HRTEM, XRD, N2 adsorption, XPS and galvanostatic cycling results suggest that confined SnO2 particles undergo an enhanced sodium alloying/dealloying process due to their special confinement inside the pores, which increases their conductivity, facilitates the diffusion of Na+ ions and buffers the large volumetric changes during charge/discharge. These high performances cannot be delivered when SnO2 is not confined and not well dispersed in the carbon pores. This work demonstrates that nano-confinement of anode species in carbon is a valuable concept affording the modification of the fundamental properties of guest species along with their electrochemical performances leading to highly stable and performing materials with a long life for Na-ion batteries.

 Please wait while we load your content...
Please wait while we load your content...