Chemical sensing in two dimensional porous covalent organic nanosheets†
Abstract
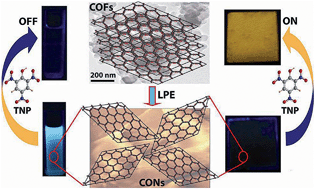
Two new imide-based crystalline, porous, and chemically stable covalent organic frameworks (COFs) (TpBDH and TfpBDH) have been successfully synthesized employing solvothermal crystallization route. Furthermore, thin layered covalent organic nanosheets (CONs) were derived from these bulk COFs by the simple liquid phase exfoliation method. These 2D CONs showcase increased luminescence intensity compared to their bulk counterparts (COFs). Notably, TfpBDH-CONs showcase good selectivity and prominent, direct visual detection towards different nitroaromatic analytes over TpBDH-CONs. Quite interestingly, TfpBDH-CONs exhibit a superior “turn-on” detection capability for 2,4,6-trinitrophenol (TNP) in the solid state, but conversely, they also show a “turn-off” detection in the dispersion state. These findings describe a new approach towards developing an efficient, promising fluorescence chemosensor material for both visual and spectroscopic detection of nitroaromatic compounds with very low [10−5 (M)] analyte concentrations.
- This article is part of the themed collections: Celebrating 15 years of exceptional research in Chemical Science and Celebrating the Chemical Science in India - Leaders in the Field Symposium

 Please wait while we load your content...
Please wait while we load your content...