One-step template-free electrodeposition of novel poly(indole-7-carboxylic acid) nanowires and their high capacitance properties†
Abstract
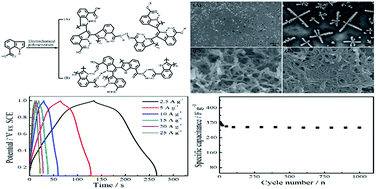
Poly(indole-7-carboxylic acid) (PICA) nanowires with conductivity of 5 × 10−2 S cm−1 were prepared by a facile, one-step and template-free electrodeposition method. The hydrogen bond interactions between the N–H group and the carboxyl group facilitated the formation of PICA nanowires. The diameter of the PICA nanowires was about 40 nm confirmed by scanning electron microscopy. Fourier transformation infrared spectroscopy and 1H NMR spectroscopy confirmed that the polymerization occurred at the C2 and C3 position on the indole ring. The electrochemical capacitance properties of the PICA nanowires were investigated with cyclic voltammetry, galvanostatic charge–discharge, and electrochemical impedance spectroscope techniques. A remarkable specific capacitance of 373.2 F g−1 was obtained at a current density of 2.5 A g−1 in 1.0 M H2SO4 solution. PICA nanowires presented an excellent cycle life with 91% specific capacitance retention after 1000 charge–discharge processes. The energy density of the symmetric full cell based on two PICA electrodes was 7.03 W h kg−1 at a power density of 4500 W kg−1. These results implied that the PICA nanowires will be a promising electrode material for supercapacitors.

 Please wait while we load your content...
Please wait while we load your content...