Fragile X mental retardation protein interactions with a G quadruplex structure in the 3′-untranslated region of NR2B mRNA†
Abstract
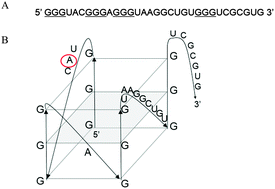
Fragile X syndrome, the most common cause of inherited intellectual disability, is caused by a trinucleotide CGG expansion in the 5′-untranslated region of the FMR1 gene, which leads to the loss of expression of the fragile X mental retardation protein (FMRP). FMRP, an RNA-binding protein that regulates the translation of specific mRNAs, has been shown to bind a subset of its mRNA targets by recognizing G quadruplex structures. It has been suggested that FMRP controls the local protein synthesis of several protein components of the post synaptic density (PSD) in response to specific cellular needs. We have previously shown that the interactions between FMRP and mRNAs of the PSD scaffold proteins PSD-95 and Shank1 are mediated via stable G-quadruplex structures formed within the 3′-untranslated regions of these mRNAs. In this study we used biophysical methods to show that a comparable G quadruplex structure forms in the 3′-untranslated region of the glutamate receptor subunit NR2B mRNA encoding for a subunit of N-methyl-D-aspartate (NMDA) receptors that is recognized specifically by FMRP, suggesting a common theme for FMRP recognition of its dendritic mRNA targets.

 Please wait while we load your content...
Please wait while we load your content...