Synthesis of metal–organic frameworks in water at room temperature: salts as linker sources†
Abstract
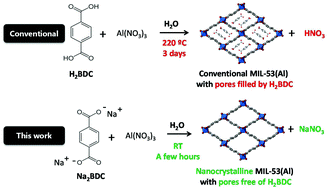
Exponential growth in the interest in MOF (Metal–Organic Framework) materials in both scientific and industrial spheres has been flourishing in the last fifteen years. However, certain environmental and economic limitations could slow down their industrial establishment. Here we present an innovative, widely applicable, straightforward, rapid, inexpensive and environmentally friendly method to prepare high-quality carboxylate-based MOFs with properties rarely obtained under conventional conditions. The method is based on the use of organic salts (instead of their homologous protonated organic ligands) as anionic linker sources, so that their solubility and the deprotonation steps, which necessarily take place in the formation of MOFs, are notably favoured in aqueous solution. As an example, the preparation of X-MIL-53-Al (X = none, NH2, NO2) materials is reported at room temperature, with the following additional advantages for some applications: they (i) are nanocrystalline, (ii) have notable and ordered inter-crystalline mesoporosity, and (iii) contain a reduced or even a negligible amount of unreacted linker within the pores. To the best of our knowledge, this is the first preparation of a 3-D highly-porous MOF under these two industrially-demanded synthesis conditions: room temperature and water as the sole solvent. The scarce commercial availability of these linker salts is a limitation that can be easily circumvented by replacing the salts with their protonated analogue plus a base. Following this method, a high-quality MOF-74 material, so far only prepared in organic solvents, could be also prepared in water and at room temperature. Even in organic media, in which this approach could be a priori unfavourable from a linker solubility point of view, MOFs with properties unattainable through conventional methods of synthesis can be prepared following this approach. That is the case for the synthesis of MOF-5 with the highest interpenetration degree ever described by just replacing terephthalic acid with disodium terephthalate as the linker source and maintaining the otherwise conventional conditions including the use of DMF as the solvent.
- This article is part of the themed collection: 2015 most accessed Green Chemistry articles

 Please wait while we load your content...
Please wait while we load your content...