Two types of novel tetra-iron substituted sandwich-type arsenotungastates with supporting lanthanide pendants†
Abstract
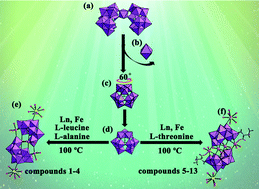
Two classes of novel tetra-iron substituted sandwich-type arsenotungastates (ATs) with supporting lanthanide (Ln) pendants KNa2 [Ln(H2O)7][Fe4(H2O)10(B-β-AsW9O33)2]·21H2O [Ln = LaIII (1), PrIII (2), NdIII (3), SmIII (4)] and [Ln(H2O)8]2[Fe4(H2O)8(L-thr)2(B-β-AsW9O33)2]·20H2O [Ln = LaIII (5), PrIII (6), NdIII (7), SmIII (8), EuIII (9), GdIII (10), TbIII (11), DyIII (12), ErIII (13)] (L-thr = L-threonine) have been synthesized by the hydrothermal reaction of the [As2W19O67(H2O)]14− precursor with Fe3+ cations and Ln3+ cations in the presence of L-thr or L-leucine and L-alanine, and further characterized by elemental analyses, IR spectra and single-crystal X-ray diffraction. Structural analyses indicate that 1–4 display the inorganic 2-D sheet architecture constructed from tetra-iron sandwiched AT [Fe4(H2O)10(B-β-AsW9O33)2]6− fragments by bridging [Ln(H2O)7]3+ cations whereas the molecular structures of the isostructural 5–13 consist of an organic–inorganic hybrid tetra-iron substituted sandwich-type AT [Fe4(H2O)8(L-thr)2(B-β-AsW9O33)2]6− fragment and two pendant [Ln(H2O)8]3+ cations. As far as we know, 1–4 represent the rare inorganic 2-D extended ATs based on transition-metal substituted sandwich-type polyoxometalate units and Ln linkers and 5–13 are the first Fe–Ln heterometallic ATs with amino acid ligands. The solid state photoluminescence (PL) measurements of 9 and 11 have been performed at room temperature. The PL emission of 9 is mainly derived from the characteristic 5D0 → 7F2 (J = 4–0) transitions of the EuIII cations whereas the PL behavior of 11 stems from the common contribution of the 5D4 → 7FJ (J = 5–3) transitions of the TbIII ions and oxygen-to-metal (O → W) charge-transfer transitions of AT segments. The thermogravimetric (TG) analyses of 1–4 and 6–12 have been investigated.

 Please wait while we load your content...
Please wait while we load your content...