Why LiFePO4 is a safe battery electrode: Coulomb repulsion induced electron-state reshuffling upon lithiation†
Abstract
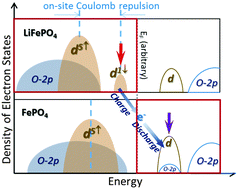
LiFePO4 is a battery cathode material with high safety standards due to its unique electronic structure. We performed systematic experimental and theoretical studies based on soft X-ray emission, absorption, and hard X-ray Raman spectroscopy of LixFePO4 nanoparticles and single crystals. The results clearly show a non-rigid electron-state reconfiguration of both the occupied and unoccupied Fe-3d and O-2p states during the (de)lithiation process. We focus on the energy configurations of the occupied states of LiFePO4 and the unoccupied states of FePO4, which are the critical states where electrons are removed and injected during the charge and discharge process, respectively. In LiFePO4, the soft X-ray emission spectroscopy shows that, due to the Coulomb repulsion effect, the occupied Fe-3d states with the minority spin sit close to the Fermi level. In FePO4, the soft X-ray absorption and hard X-ray Raman spectroscopy show that the unoccupied Fe-3d states again sit close to the Fermi level. These critical 3d electron state configurations are consistent with the calculations based on modified Becke and Johnson potentials GGA+U (MBJGGA+U) framework, which improves the overall lineshape prediction compared with the conventionally used GGA+U method. The combined experimental and theoretical studies show that the non-rigid electron state reshuffling guarantees the stability of oxygen during the redox reaction throughout the charge and discharge process of LiFePO4 electrodes, leading to the intrinsic safe performance of the electrodes.

 Please wait while we load your content...
Please wait while we load your content...