Kinetic and mechanistic study of the reaction of OH radicals with methylated benzenes: 1,4-dimethyl-, 1,3,5-trimethyl-, 1,2,4,5-, 1,2,3,5- and 1,2,3,4-tetramethyl-, pentamethyl-, and hexamethylbenzene†
Abstract
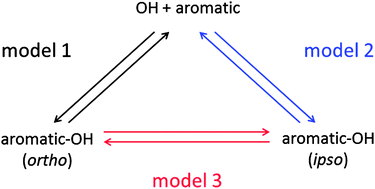
The reaction of OH radicals with a series of methylated benzenes was studied in a temperature range 300–350 K using a flash-photolysis resonance fluorescence technique. Reversible OH additions led to complex OH decays dependent on the number of distinguishable adducts. Except for hexamethylbenzene, triexponential OH decay curves were obtained, consistent with formation of at least two adduct species. For three compounds that can strictly form two adduct isomers for symmetry reasons (1,4-dimethyl-, 1,3,5-trimethyl-, and 1,2,4,5-tetramethylbenzene) with OH bound ortho or ipso with respect to the methyl groups, OH decay curves were analysed in terms of a reaction mechanism in which the two adducts can be formed directly by OH addition or indirectly by isomerization. In all cases one adduct (add1) is dominating the decomposition back to OH. The other (add2) is more elusive and only detectable at elevated temperatures, similar to the single OH adduct of hexamethylbenzene. Two limiting cases of the general reaction mechanism could be examined quantitatively: reversible formation of add2 exclusively in the OH reaction or by isomerization of add1. Total OH rate constants, adduct loss rate constants and products of forward and reverse rate constants of reversible reactions were determined. From these quantities, adduct yields, equilibrium constants, as well as reaction enthalpies and entropies were derived for the three aromatics. Adduct yields strongly depend on the selected reaction model but generally formation of add1 predominates. For both models equilibrium constants of OH reactions lie between those of OH + benzene from the literature and those obtained for OH + hexamethylbenzene. The corresponding reaction enthalpies of add1 and add2 formations are in a range −87 ± 20 kJ mol−1, less exothermic than for hexamethylbenzene (−101 kJ mol−1). Reaction enthalpies of possible add1 → add2 isomerizations are comparatively small. Because results for 1,3,5-trimethylbenzene are partly inconsistent with a direct formation of add2, we promote the existence of isomerization reactions. Moreover, based on available theoretical work in the literature, add1 and add2 are tentatively identified as ortho and ipso adducts, respectively. Total OH rate constants were obtained for all title compounds. They can be described by Arrhenius equations: kOH = A × exp(−B/T). The parameters ln(A/(cm3 s−1)) = −25.6 ± 0.3, −25.3 ± 0.6, −27.3 ± 0.3, −24.6 ± 0.3, −26.2 ± 0.4, −26.2 ± 0.4 and −24.5 ± 0.2, and B/K = −160 ± 90, −550 ± 180, −1120 ± 90, −330 ± 100, −820 ± 100, −980 ± 130, and −570 ± 40 were determined for 1,4-dimethyl-, 1,3,5-trimethyl-, 1,2,4,5-, 1,2,3,5- and 1,2,3,4-tetramethyl-, pentamethyl-, and hexamethylbenzene.


 Please wait while we load your content...
Please wait while we load your content...