Mechanistic elucidation of C–H oxidation by electron rich non-heme iron(iv)–oxo at room temperature†
Abstract
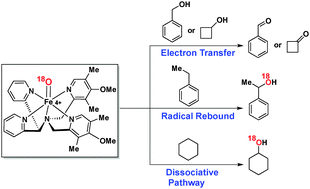
Non-heme iron(IV)–oxo species form iron(III) intermediates during hydrogen atom abstraction (HAA) from the C–H bond. While synthesizing a room temperature stable, electron rich, non-heme iron(IV)–oxo compound, we obtained iron(III)–hydroxide, iron(III)–alkoxide and hydroxylated-substrate-bound iron(II) as the detectable intermediates. The present study revealed that a radical rebound pathway was operative for benzylic C–H oxidation of ethylbenzene and cumene. A dissociative pathway for cyclohexane oxidation was established based on UV-vis and radical trap experiments. Interestingly, experimental evidence including O-18 labeling and mechanistic study suggested an electron transfer mechanism to be operative during C–H oxidation of alcohols (e.g. benzyl alcohol and cyclobutanol). The present report, therefore, unveils non-heme iron(IV)–oxo promoted substrate-dependent C–H oxidation pathways which are of synthetic as well as biological significance.

 Please wait while we load your content...
Please wait while we load your content...