Electrochemical detection of 8-hydroxy-2′-deoxyguanosine as a biomarker for oxidative DNA damage in HEK293 cells exposed to 3-chloro-1,2-propanediol†
Abstract
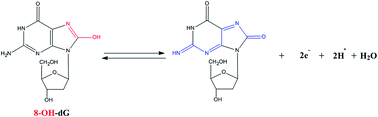
We investigate here the selective cytotoxicity of 3-chloro-1,2-propanediol (3-MCPD) on HEK293 cells by analyzing the cell growth inhibition, morphological changes, intracellular reactive oxygen species (ROS) production, and DNA damage. We further demonstrate that 3-MCPD inhibits the growth of cells and induces 8-hydroxy-2′-deoxyguanosine (8-OH-dG) generation via ROS-mediated oxidative DNA damage in HEK293 cells. To provide a detection system, we fabricated a modified electrode with poly(3-acetylthiophene) (P3AT), for electrochemical detection of 8-OH-dG in oxidation-damaged cells. By.electropolymerization using cyclic voltammetry (CV), we deposited 3-acetylthiophene (3-AT) on a glassy carbon electrode (GCE). The conducting polymer, P3AT, greatly enhances the peak current via the dramatic electrocatalytic effect on the oxidation of 8-OH-dG. We further examined the effects of pre-concentration potential, time, scan rate, and pH value on voltammetric behavior and detection of 8-OH-dG. Under optimal conditions, the anodic peak currents of differential pulse voltammetry (DPV) maintain a linear relationship with the 8-OH-dG concentration between 0.5 and 35 μM, with a correlation coefficient of 0.9963. We estimated the detection limit of 8-OH-dG to be 31.3 nM (S/N = 3). The proposed modified electrode demonstrates excellent reproducibility and stability, making it an ideal candidate for amperometric detection of 8-OH-dG. We performed the detection on real cell samples with satisfactory results.

 Please wait while we load your content...
Please wait while we load your content...