A sensitive LC-ESI-MS/MS method for the determination of clotrimazole in human plasma
Abstract
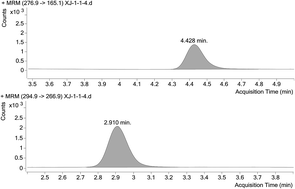
A rapid, sensitive and specific liquid chromatography-electrospray ionization-tandem mass spectrometry (LC-ESI-MS/MS) method for the determination of clotrimazole in human plasma was developed and validated. The plasma samples were extracted with a mixed solvent of methyl tert-butyl ether–dichloromethane (4 : 1, v/v), and estazolam was selected as the internal standard. Then, the separation was carried out on a Phenomenex Luna CN column (2.0 × 150 mm, 5 μm), using a mobile phase of methanol-0.1% aqueous formic acid solution (85 : 15, v/v). A triple quadrupole mass spectrometer with the positive ionization mode was used for the determination of target analytes. The monitored ion transitions were m/z 276.9 → 165.1 for clotrimazole and m/z 294.9 → 266.9 for estazolam, respectively. The calibration curve of clotrimazole was established over the range of 0.01563–1.000 ng mL−1 (r2 = 0.9935). The intra- and inter-day precisions were less than 10% and all the biases were not more than 9%. The mean extraction recovery of clotrimazole was greater than 68.4% and no significant matrix effect was detected. The LLOQ of 0.01563 ng mL−1 is sensitive enough to perform pharmacokinetic studies after clotrimazole administration.

 Please wait while we load your content...
Please wait while we load your content...